
When the rock hits the ground, this energy, in turn, is transformed into internal energy of the rock and the ground in the vicinity of the impact the molecules The initial potential energy of the rock changes to kinetic energy as the rock falls. Yet it does not happen spontaneously.Īs a second example, consider what happens when you drop a rock and it hits the ground. If heat were to leave the colder object and pass to the hotter one, energy could still be conserved. For example, when a hot object is placed in contact with a cold object, heat flows from the hotter one to the colder one, never spontaneously from colder to hotter. There are, however, many processes we can imagine that conserve energy but are not observed to occur in nature. The first law of thermodynamics states that energy is conserved. 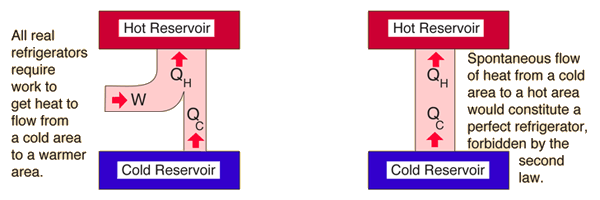
Examples of the second law of thermodynamics To explain this lack of reversibility scientists in the latter half of the nineteenth century formulated a new principle known as the 2nd law of thermodynamics. 
The first law of thermodynamics would not be violated if any of these processes occurred in reverse.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
